Codon Digest: Ghana Approves A Better Malaria Vaccine
Plus: A better way to edit genes in the brain and gene-edited bananas bypass GMO regulations.
Codon Digest is a weekly roundup of amazing things in biology and technology. This newsletter’s format has shifted again, as I try to figure out the most enticing way to present this stuff. Please send me your feedback.
🔥 Five Amazing Things
(That happened this week…)
Genome editing in the mouse brain with minimally immunogenic Cas9 RNPs. By Elizabeth C. Stahl et al. on bioRxiv.
If you want to edit a gene inside of a neuron, what do you do? Most people would take the two CRISPR gene-editing components (a Cas9 protein and guide RNA), package them up inside of a virus, and then inject the viruses into the skulls of mice. These viruses enter the brain and deliver the gene-editing ingredients into neurons. Unfortunately, they can also trigger immune responses, and they are not super efficient at gene-editing some parts of the brain.
A new study from Jennifer Doudna’s lab shows that ribonucleoproteins (basically little balls of Cas9 and guide RNAs, packaged inside a fat bubble) can edit genes in the brain better than viruses. These fat bubbles are injected straight through the skull. In one experiment, they successfully edited 14% of neurons near the injection site, which was sufficient to reduce symptoms in a mouse model of a neurological disorder, called fragile X syndrome.
These ribonucleoproteins also caused a lower immune response compared to the viruses, and are much easier to make in the lab. Delivering a 25μM dose of the Cas9 ribonucleoproteins led to lower levels of vehicle-specific antibodies 90 days post-injection, compared to the viruses, and also did not cause T-cell gene signatures to spike up.

Promising new malaria vaccine for kids approved in Ghana. Associated Press. Read
Ghana is the first country to approve a new malaria vaccine developed at the University of Oxford. This is not the first vaccine approved to prevent malaria, but it is by far the most effective. A clinical trial from Burkina Faso, published last year, showed that this vaccine was up to 80% effective at preventing malaria (compared to an existing vaccine distributed by the WHO, called Mosquirix, which is about 30% effective). The vaccine, called R21/Matrix-M, is made by fusing a protein secreted by the malaria parasite to a hepatitis B surface antigen that helps the vaccine enter cells.
The African nation approved the vaccine for use in children between 5 and 36 months of age. This vaccine is being “scaled up” at the Serum Institute of India, which “says it could produce up to 100 million doses depending on demand.” Malaria currently kills about 500,000 children every year; enough to fill nearly 7,000 school buses. That is a sobering thought.

Tropic’s non-browning gene-edited banana cleared for production in the Philippines. Press release.
In a recent essay, I explained how a U.K. company, called Tropic Biosciences, had developed bananas that stay yellow for more than a month on grocery store shelves. It’s a remarkable achievement, with the potential to save billions of bananas from going to landfill each year. The company estimates that its fruit could reduce carbon emissions across the banana supply chain by 25 percent.
These gene-edited bananas were submitted for regulatory approval in the Philippines (a top five global exporter of bananas) sometime last year. The country has now ruled that the gene-edited bananas are exempt from GMO regulations because they do not carry additional DNA sequences, and so they will not have to pass through strict safety testing. “With this determination the Tropic non-browning can be freely imported and propagated in the Philippines,” according to a company press release.
(Side note: The man who developed the Cavendish banana, the main banana we all eat today, was a Brit named Sir Joseph Paxton. He cultivated the bananas “in state-of-the-art greenhouses he'd designed in Derbyshire for the duke of cavendish.” Thanks to Tom Ellis for sharing.)
Read my essay here.
Targeting Sex Determination to Suppress Mosquito Populations. Ming Li et al. in bioRxiv.
Mosquitoes have killed more humans than any other animal (even sharks, my great nemeses.) They spread Zika, malaria, dengue, and chikungunya. Genetic engineering can be used to eradicate mosquitoes in two ways: Through gene drives or the “sterile insect technique.”
The former is a controversial tool that uses clever bioengineering to propagate lethal genes through a species, thus causing a population to collapse. Gene drives are difficult to control, and none have been tried in the wild. The sterile insect technique, by contrast, has already been used in lots of places. You basically x-ray a bunch of male mosquitoes to make them sterile, and then release them en masse into nature. The males fly around, breed with females, and produce eggs that never hatch. The population plummets. This is the method that Oxitec, a UK company, has been using to curb mosquitoes in the Florida Keys and parts of Brazil.
Unfortunately, the sterile insect technique is tedious and slow. X-rays are horribly inefficient. Some males become sterile, but others don’t. A new study has improved upon this process, using CRISPR gene-editing to make males sterile with very high efficiencies. When sterile males were released into a cage with females at a 20:1 ratio (which is quite a lot), the population went extinct after about five generations. Not all gene-edited males were 100% sterile, but these results may be a more promising means to control mosquitoes in the wild.

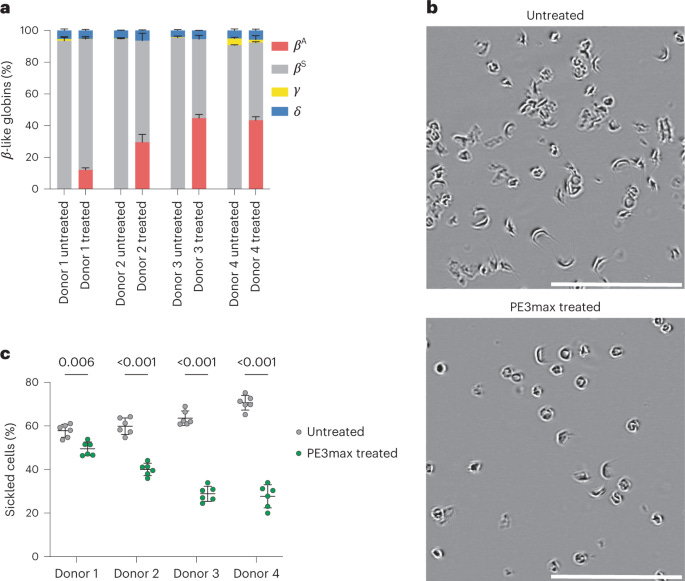
Ex vivo prime editing of patient haematopoietic stem cells rescues sickle-cell disease phenotypes after engraftment in mice. Everette K.A. et al. in Nature Biomedical Engineering.
Sickle-cell disease is a genetic disorder that affects an estimated 100,000 Americans (and 1-in-365 Black Americans). It is caused by a mutation in the β-globin gene, which leads to abnormally shaped red blood cells that can pool in the spleen and block blood vessels. A normal red blood cell lives for 120 days; sickle cells live for 10-20 days.
Red blood cells are made by stem cells in the bone marrow. A research group at Harvard has now used prime editing — a gene-editing tool that can make small insertions or deletions in the genome — to directly fix the β-globin gene in anywhere from 15 to 41 percent of these stem cells. When the edited stem cells were transplanted back into mice, they engrafted into the bone marrow and produced healthy red blood cells after 17 weeks.
This new tool could be a “one-and-done” cure for sickle-cell disease. Right now, the only FDA-approved treatment is to transplant blood stem cells from a healthy person into a sickle-cell patient, but some people never find a suitable donor. This new paper would solve that problem because no donors are needed — the stem cells come from the patient’s own body.
See this Twitter thread for more details.
This is the future I want: Live in a van and roam the world. Engineer microbes while sitting upon my twin-sized mattress. By putting labs into vans, we could democratize access to biotechnology. We could bring educational lessons to kids in rural areas, and expand the field to borders far outside of Boston and San Francisco.
📎 Papers
Strands of DNA can be used to build molecular logic gates. This paper shows how to build every type of gate — NAND, NOT, NOR, and so on — using DNA, and even includes an open-source software to design everything. Nature Communications. Read
This paper presents a new way to get DNA into skeletal muscle by engineering fusogen proteins, which help viruses stick to and enter human cells. Cell. Read
A suite of calcium sensors, called NEMO, are way brighter than any other proteins currently available to monitor neural activity. These proteins are used to “watch” neurons fire in real-time, in both mammals and plants! Nature Methods. Read
A new tool to “hypermutate” genes inside living cells, at a rate of roughly 5 mutations every 1,000 bases. Nucleic Acids Research. Read
Engineered microbes could be used to build biomaterials, and recycle atoms, on the Moon or Mars. This is a beautiful primer to the field of space synthetic biology. Nature Communications. Read
Every living cell uses DNA as its genetic material, and DNA itself is made up of individual molecules (like A, T, C, and G.) But what happens when cells are engineered so that they can’t make their own DNA, but are instead “fed” the raw molecules? Well, they grow a bit slower, and they look a bit odd, but they still survive. eLife. Read
📰 In the News
AI will change how biology experiments are done. Most pipetting will become automated. Machines will be programmed with little more than our voices. “It is time now to begin working on what the laboratory of the future will look like: how we can make it as productive as possible, and how we can make sure that it remains safe and controllable in the years to come,” writes Sam Rodriques. Read
UC Berkeley scientists Jennifer Doudna and Jill Banfield got $70 million in funding to edit microbes, inside cow stomachs, to reduce methane emissions. Fierce Biotech. Read
Interestingly, a startup called Native Microbials recently won $1.4 million to do something similar.
Fertility gets little attention from federal funders. But “a new crop of biotech startups” are pioneering methods to make gametes (sperm or eggs) from stem cells, and to enable same-sex couples to produce biological offspring. The New Yorker. Read
A few months ago, a study in Nature suggested that synonymous mutations — changes in DNA that do not change the amino acid sequence of a protein — were deleterious. That is, they actively hurt living organisms. The study was debunked recently, and the whole saga offers an intriguing window into just how much a prestigious journal can neuter criticisms. Twitter thread. Read
A gene therapy for Duchenne muscular dystrophy, developed by a company called Sarepta, is expected to be approved (or rejected) by the F.D.A. by May 29th. The F.D.A. was leaning toward rejecting the gene therapy earlier this year, according to STAT reporting, and then a “top official intervened.” STAT. Read
Roundworms fed cannabinoids get the munchies, according to a study that was published on 4/20. Ars Technica. Read
A profile of Tracie Seimon, a scientist who uses DNA sequencing to detect and monitor wild species in “Peru, Myanmar, Vietnam, Cambodia, Russia, Uganda and Rwanda.” Quanta Magazine. Read
🧠 Musings
For the first time, scientists have read a text authored by Ptolemy, the renowned Roman astronomer and mathematician. Ars Technica. Read
People say that synthetic biology doesn’t scale. That may be (mostly) true over the last couple decades, but it shouldn’t stop us from imagining entire buildings powered by photosynthetic algae or patio furniture woven from materials produced from yeast. A new review imagines this implausible future. Rachel Armstrong. Read
An upcoming, week-long hackathon will challenge attendees to build products at the intersection of synthetic biology and games. Sounds interesting and registration is free. Two days are in San Francisco. Read
This looks like a useful Python tool to analyze genomics data in Jupyter notebooks. Read
📈 Companies
Microbes sold by Pivot Bio, a company that uses living organisms to replace the nitrogen in synthetic fertilizers, “were used on more than 3 million acre[s] of U.S. cropland in 2022 — 300% year-over-year growth.” Press release. Read
AI tools can now design never-before-seen proteins. But how do you know whether they’ll work in the real world? A new startup, called Adaptyv Bio, is building a microfluidics platform to build and test lots of proteins at once, using “1,000 times fewer reagents” than any existing alternatives. This is very important. Looks like a cool company. Labiotech.eu. Read
Moderna is using quantum computers, developed by IBM, to design future therapeutics. Fierce Biotech. Read
Cultivarium, a company building molecular tools for non-model organisms (think everything but E. coli and yeast), signed an agreement with the nonprofit, ATCC, to help get their microbes to researchers. This could be an important step toward democratizing access to extremophiles and other cool bugs. Press release. Read
Complement Therapeutics, a company building gene therapies for vision-related diseases, raised €72 million in Series A funding. Press release. Read
Eli Lilly is adding 200 jobs at its drug manufacturing plants in Indiana, which will cost $3.7 billion together. Press release. Read
Thanks for reading,
— Niko McCarty
Disclosure: The views expressed in this blog are entirely my own and do not represent the views of any company or university with which I am affiliated.




Great digest. Loads of cool news. Love seeing the improvement in Childhood malarial rates in Africa, but certainly more work to be done. Also, the Lab Van idea would be pretty sweet. I would love to see some initiative pull together resources for maybe a lab RV that could do the sci-comm outreach to rural or low income areas. And about the nitrogen fixing soil bacteria... I think we're in for a lot more from that after reading about the US Biotech investment strategy. Very excited for that stuff.
Great newsletter. However, I'd just like to clarify whether it's Ghana or Uganda (as the title mentions) that approved the new vaccine